Investigation the potential of nanocarriers in gene therapy
Certain cancers and related nanocarriers factors for targeted gene therapy.
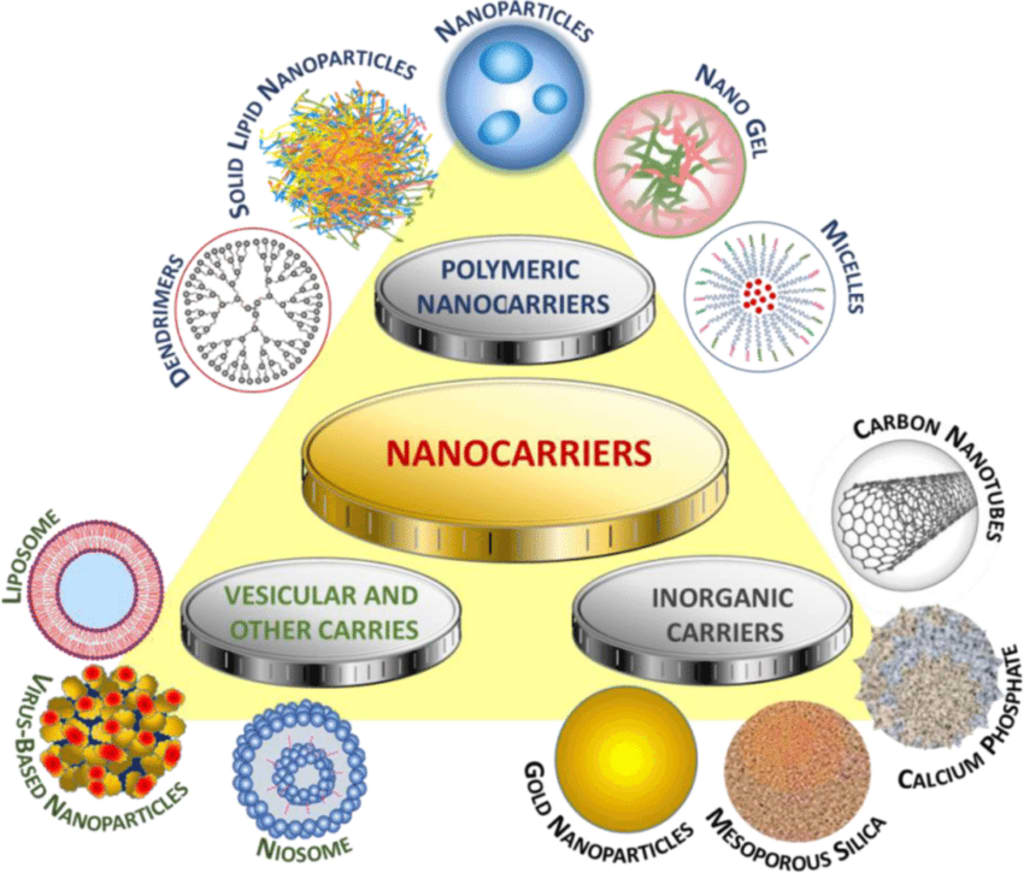
Certain cancers and related nanocarriers
In all cases, tumours are primarily brought on by mutations or other defects in the tumour suppressor genes, which prevent cell growth and promote programmed cell death (apoptosis), or in the proto-oncogenes, which control cell proliferation and differentiation. The development of malignant cells with their distinct characteristics of unchecked cell growth, inability to stop excessive cell division, lack of apoptosis, and capacity to penetrate nearby and distant tissues is caused by these altered genes. Radiation, chemical agents, such as carcinogens, physical irritants, hereditary factors, and viruses are risk factors for genetic alterations. One of the most deadly diseases in the world, tumours are on the rise, with more than 10 million new cases reported each year.

Breast tumour:
Doxorubicin, paclitaxel, and cisplatin are just a few of the chemotherapeutic drugs that are utilised to treat breast tumours in both neoadjuvant and adjuvant therapy techniques. The first-line treatment for breast tumours is doxorubicin. By blocking DNA intercalation and macromolecular production inside tumour cells, it slows the growth of tumour cells. Due to the doxorubicin's toxicity with increasing dose, the main side effects connected with it are hair loss, an increase in neutrophils, and heart issues. Breast tumours are typically treated with platinum medicines, such as cisplatin or carboplatin, in combination with other anticancer medications. By maintaining DNA binding and cross-linking, cisplatin causes apoptosis, which stops the proliferation of tumour cells.
Lung tumour:
Gold nanoparticles with sensors were successfully created for this purpose and utilized to identify the lung tumour. In a highly humid environment, they were used to distinguish between the breath of lungs tumour patients and that of healthy people. These sensor-based NPs were low-cost, non-invasive lung tumour diagnostic tools. Similar to this, 200 nm average particle size PLGA-based anticancer medication (TAS-103)-loaded NPs were created, which had an improved antitumor activity in lungs tumour. Additionally, poly(butylcyanoacrylate) NPs with doxorubicin in them were successful in treating lung tumours. In order to target lung tumours, liposomes were also employed as nanocarriers for various medications. For p53 tumour suppressor gene-mediated increased cytotoxicity in A549, a multicomponent liposomal formulation containing two cytotoxic drugs (etoposide and docetaxel) was created.
Investigating the potential of nanocarriers in targeted gene therapy
Targeted gene therapy is one of the many applications that nanocarriers, which are specialized delivery systems at the nanoscale, have the potential to be extremely useful for. These nanoscale vehicles offer a platform for regulated encapsulation and delivery of therapeutic substances, such as genes, medications, or diagnostic agents, to certain cells or tissues. By utilising the special qualities of nanoparticles, nanocarriers provide a number of benefits over conventional delivery techniques, creating new opportunities for precise and effective treatment procedures.
The development of nanocarriers has been extremely beneficial for the science of gene therapy, which tries to treat diseases by modifying gene expression or correcting genetic abnormalities. The possibilities for gene-based therapies have been transformed by their capacity to shield genetic material from deterioration, enhance stability, and enable targeted delivery. Nanocarriers act as protective barriers that allow therapeutic genes to be delivered to target cells in a safe and effective manner while navigating numerous physiological obstacles within the body.
Different nanomaterials, such as liposomes, polymeric nanoparticles, dendrimers, or viral vectors, each with unique properties and capacities, can be used to create nanocarriers. To improve their targeting specificity, these nanoscale carriers can be altered and functionalized with surface ligands, antibodies, or peptides. The treatment efficacy can be increased and off-target effects decreased by using nanocarriers to target precise interactions between target cells and desired places.
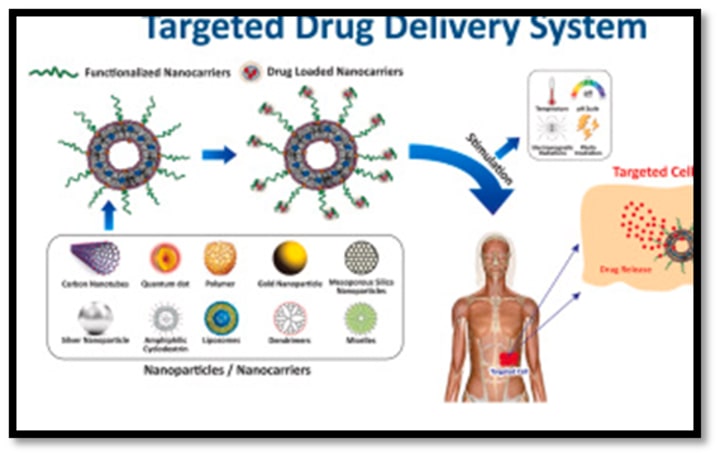
Therapeutic genes are enclosed in nanocarriers, which not only prevent genetic material from deterioration but also allow for a regulated release at the target spot. This controlled release can be customised to produce long-lasting and ideal therapeutic effects, extending the presence and boosting the activity of therapeutic genes.
Additionally, nanocarriers can be developed to have good biocompatibility, biodegradability, and low immunogenicity, which reduces the likelihood of adverse effects and enhances overall safety profiles. To ensure their suitability with biological systems, extensive study is being done to assess their immunological and toxicological responses.
Preclinical studies have shown the promise of nanocarriers for targeted gene therapy, with encouraging outcomes in the treatment of a range of illnesses, including genetic abnormalities, cancer, and cardiovascular ailments. However, more study is required to increase their efficiency of administration, optimise their design, and handle problems like large-scale production and regulatory issues for clinical translation.
As a result of their special qualities and abilities, nanocarriers have demonstrated significant promise for targeted gene therapy. To cure genetic abnormalities or other diseases, gene therapy involves inserting therapeutic genes into a subset of cells. However, a number of internal barriers in the body make it difficult to transport genetic material to the target cells. By encasing and safeguarding the therapeutic genes, enhancing their stability, and enabling their targeted delivery, nanocarriers offer a viable option.
Factors for targeted gene therapy:
When looking into the potential of nanocarriers for targeted gene therapy, the following important factors should be taken into account:
1. Effective encapsulation: Therapeutic genes should be able to be effectively encapsulated by nanocarriers to prevent degradation and improve their stability while in circulation. Various encapsulation techniques are available on various nanocarrier platforms, including liposomes, polymeric nanoparticles, and viral vectors.
2. Targeting techniques: Nanocarriers can be made to target particular cells or tissues, increasing the selectivity and effectiveness of gene therapy. Nanocarriers can recognise and bind to particular cell receptors thanks to surface modifications such ligand conjugation or antibody targeting, which improves their internalisation and uptake.
3. Biocompatibility and safety: It's crucial to make that nanocarriers are safe, non-toxic, and suitable for clinical use. To assess any potential negative consequences, such as immunological reactions, cytotoxicity, or off-target effects, in-depth investigations should be carried out.
4-Stability and controlled release: Until they reach the target site, nanocarriers should keep their structural integrity and safeguard the genetic material. Additionally, to ensure sustained and regulated gene delivery and guarantee the therapeutic genes are released over the proper duration, controlled release mechanisms can be included into the nanocarriers.
5- In vivo performance: To comprehend the behaviour, distribution, and effectiveness of nanocarriers, it is essential to assess their in vivo performance. The biodistribution, pharmacokinetics, and therapeutic effects of the nanocarriers in pertinent disease models can be evaluated using animal models
6- Translational considerations: Scalability, manufactureability, and regulatory requirements for clinical translation should all be taken into account while researching nanocarriers for targeted gene therapy.
Future Perspective:
The development of new-generation nanocarrier medications is one of the major obstacles to current advancements in nanotechnology being employed effectively for the treatment of various tumours. By interacting with the receptors on the chosen cells and tissues as well as the surface-attached ligand, this expansion would validate the energetic targeting of tumours. But there are challenges to be solved, including a lack of adequate knowledge, difficulty in piercing the cell membrane, a limited therapeutic window for medications, regulatory challenges, and cost-effectiveness.
Only a few of these promising preclinical drug delivery systems have made it to market, despite the fact that researchers have explored and created a number of innovative drug delivery methods to increase treatment efficiency in patients. The crucial gaps in the transformation of anticancer drug-loaded nanocarriers may be to blame for this. To get around these issues, it is crucial to change some of the established models. In this regard, extremely complex efforts are needed to quickly fix a few problems in order to achieve the safe use of freshly produced nanocarriers in clinical investigations. These involve the creation of typical nanoformulations that have had their efficacy, safety, and potential toxicities tested both in vitro and in vivo.
Conclusion
One of the most recent strategies for the delivery of tumour drugs is nanotechnology. We covered a wide range of nanocarriers that are being employed as cutting-edge treatments for various tumours. These nanocarriers have revolutionised the way that cancer drugs are delivered since they specifically target tumours with enriched permeability and withhold effect as much as is necessary. The hope of individuals battling tumours has been boosted by these exciting developments in cancer therapy and the groundbreaking creation of various novel medication delivery systems. Future drug dosage management is anticipated to increase the usage of nanocarrier systems for the administration of anticancer drugs while also minimising side effects and maximising systemic drug release from the nanocarriers.
As a strong tool for targeted gene therapy, nanocarriers hold enormous promise for precise and successful therapies. The field of medicine could undergo a revolution as a result of their capacity to encapsulate, safeguard, and distribute therapeutic genes to particular cells or tissues, enabling customised interventions and better patient outcomes. Future applications of nanocarrier technology in therapeutic settings will be made possible by rigorous scientific research and ongoing technological breakthroughs.





Comments
There are no comments for this story
Be the first to respond and start the conversation.